By, Eric Abel, PhD, MSc, FLASH Solution Architect at Varian Medical Systems
I presented at the PTCOG annual meeting in Manchester on the techniques employed to enable the first ever FLASH irradiations with a ProBeam® system, performed on mice in a non-clinical mode, at the Maryland Proton Treatment Center (MPTC). FLASH therapy is a non-invasive treatment using an external beam delivered in ultra-high doses, at ultra-high speeds (less than 1 second), and in one to three sessions. FLASH represents an exciting and potentially promising new direction in the treatment of cancer.
I would like to acknowledge and thank our partners at the University of Maryland School of Medicine and MPTC for their participation and help in this preclinical study.
The Global Translational Science (GTS) study was motivated by a report by Favaudon et al. [1] at Institut Curie, in which significant normal tissue sparing with comparable tumor control was reported for mice treated with whole thoracic irradiation at FLASH vs conventional dose rates. The studies by the Curie group were all performed using electrons from a customized linac, with a beam energy and size that were not conducive to clinical translation. The primary objective of the GTS study, therefore, was to repeat the Favaudon experiment but where the only parameter that was changed was the irradiation mechanism, and thereby investigate the feasibility of FLASH on the ProBeam® system, with the intent of exploring a shorter time horizon over which this exciting treatment paradigm could be translated into the clinic.
The GTS experiment began in the fall of 2016 at the University of Maryland School of Medicine with roughly 400 mice total, and with groups of 10 mice irradiated simultaneously (Figure A below). Additionally, requirements were imposed by the biologists that provided constraints on lung coverage as well as dose limits to adjacent organs (Figure B below).
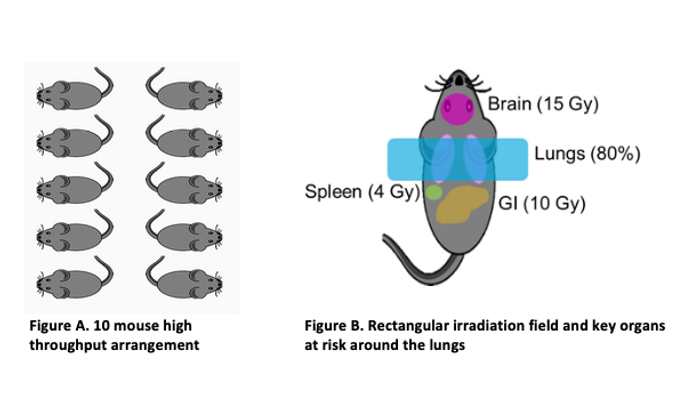
Meeting these requirements in the high throughput setup meant designing an irradiation field that would be robust to mouse alignment, breathing and variations in mouse size. A quick review of a subset of mouse CT scans revealed a high level of uniformity in mouse anatomical geometry, thereby reducing the problem to alignment and breathing.
To generate the field designs, a MOBY™ digital mouse phantom from Duke University was imported into the Eclipse™ 13.7 treatment planning system, where the organs were segmented and an initial rectangular array of uniform spots was generated (Figure C, upper right). The resultant dose was calculated and evaluated against the dose constraints. Based on the comparison, the field was translated or the spot spacing was adjusted until all constraints were matched. As a second check, the mouse phantom was also imported into the TOPAS Monte Carlo tool.
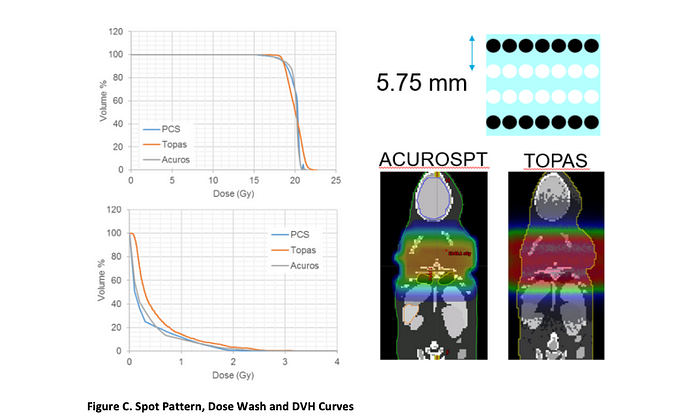
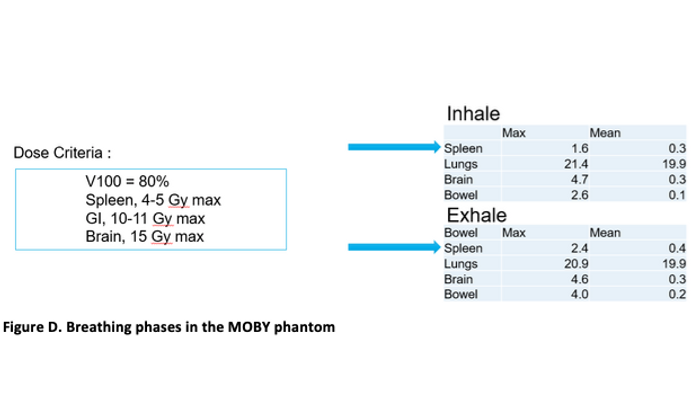
Figure C. summarizes the final spot pattern, as well as the resulting dose wash comparison and DVH curves for TOPAS and the Eclipse analytical (PCS) and Monte Carlo (AcurosPT) calculations. The robustness of the field against breathing motion was evaluated by calculating the dose to all organs for the extreme (inhale, exhale) breathing phases in the MOBY phantom. The table below (Figure D) shows that all constraints were met for both phases.
Finally, the sensitivity to mouse alignment was evaluated by using the robustness tool in Eclipse for shifts up to 2 mm in each direction. This analysis informed a 1 mm bias of the field in the cranial direction to spare dose to the spleen, with a resultant 20% under-dosing of the lowest lobe of the lung. While the lung coverage was not ideal, it was deemed more acceptable than excess spleen dose.
In the experimental setup, the mouse alignment was achieved by first roughly placing the mouse sternum on a groove which was machined into a plastic carriage tray. The mouse position was then refined through repeated radiographs of the mouse using the ProBeam system’s onboard imagers to ensure proper relative alignment of the mouse lung with the machined groove. Statistical analysis of the mouse position post-irradiation validated that all mice were positioned within the alignment window specified by the robustness analysis.
The results of the GTS study were reported in a poster presentation by Girdhani et al. [2] at the American Association for Cancer Research conference in January 2019, with a pending peer reviewed publication currently in process.
* FLASH therapy is under preclinical research. It is not available for clinical use and there is no guarantee of future commercialization.
Video of the presentation at PTCOG
- Favaudon V, Caplier L, Monceau V, et al. Ultrahigh dose-rate FLASH irradiation increases the differential response between normal and tumor tissue in mice. Sci Transl Med. 2014 Jul 16;6(245):245ra93. doi: 10.1126/scitranslmed.3008973.
- Girdhani et al. FLASH: a novel paradigm changing tumor irradiation platform that enhances therapeutic ratio by reducing normal tissue toxicity. Global Translational Sciences, Varian Medical Systems, Palo Alto CA; Department of Radiation Oncology, University of Maryland, Baltimore, Maryland